Cancer cells divide and multiply rapidly within the tumour; these cancer cells carry highly electrically charged elements, which play a significant role in cell division.1 Tumour Treating Fields (TTFields) are a non-invasive anticancer treatment that utilizes low-intensity (1–3 V/cm), intermediate frequency (100–300 kHz), alternating electric fields delivered through transducer arrays placed on the skin around the tumour region to physically disrupt cancer cell division and eventually lead to apoptosis.2
The antimitotic effect of TTFields is prominent in rapidly diving cancer cells, sparing quiescent cells, and is also dependent on the intensity, frequency, and direction of the applied electric fields.2,3 Research suggests that a maximal cytotoxic effect is observed in a range of cell types when they are treated with TTFields at a frequency between 150 and 200 kHz, and that frequencies of less than 50 kHz or greater than 500 kHz have minimal cytotoxic effect.4
These properties of TTFields suggest that such modality may be useful in treating multiple cancer types by alternating the frequency and intensity of TTFields.3 Evidence suggests that electrical fields should be applied in parallel to the axis of cell division to obtain an optimal effect.2
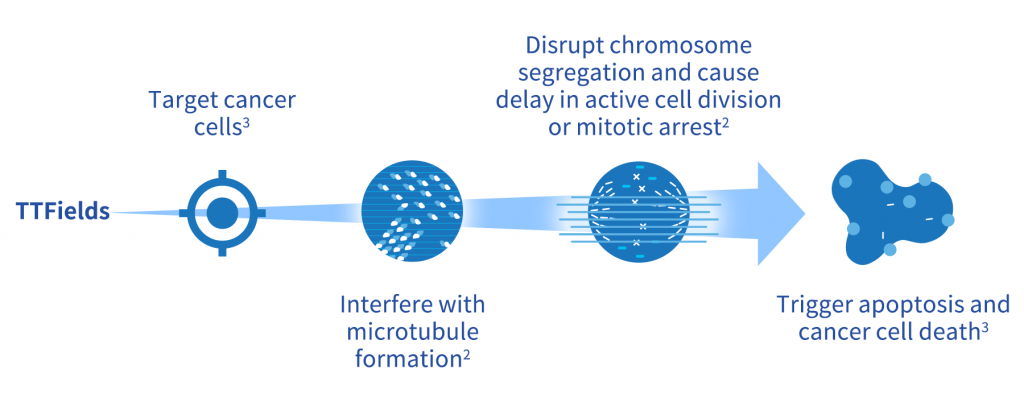
Mechanism of action
Used with Permission From Novocure.
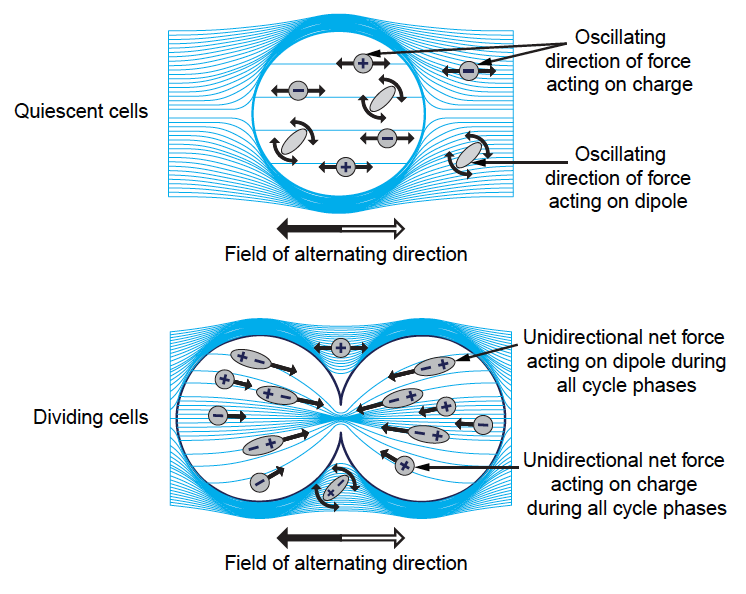
The mechanism of action of TTFields on mitosis can be explained by two fundamental electrokinetic phenomena – Dipole alignment and dielectrophoresis.5
Dipole alignment depicts a situation when electric fields exert directional forces on proteins with high intramolecular dipole moments (defined as a difference in electronegativity) such as tubulin dimers and septins.5,6 Dipole alignment disrupts normal mitotic spindle formation during metaphase, leading to improper chromosome segregation.5 During anaphase, the positioning of the cytokinetic furrow were also disrupted, which subsequently leads to mitotic arrest.5
Dielectrophoresis describes a phenomenon when polar components are pulled to an area of the highest field intensity within a non-uniform electric field.5 In the context of mitosis, during telophase, the formation of the hourglass shape creates a non-uniform electric fields within the cell, pulling charged macromolecules and organelles to the mitotic furrow, where the field intensity is the highest.5,6 This subsequently leads to disruption of cell segregation, resulting in mitotic exit and cell apoptosis.5
Dipole alignment and dielectrophoresis in quiescent and dividing cells6
Pre-clinical findings
The first piece of in vitro evidence of TTFields was reported in 2004 by Kiron et al.3 The application of low-intensity (1–3 V/cm), intermediate-frequency (100–300 kHz), alternating electric fields inhibited cell proliferation in multiple human and rodent tumour cell lines.6
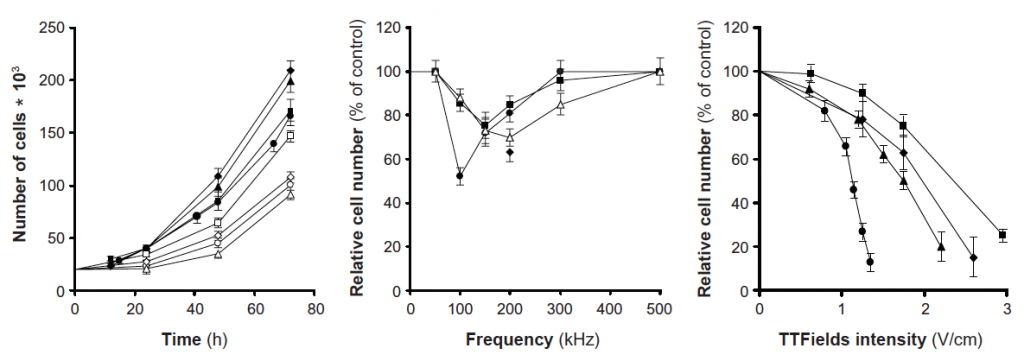
In vitro results suggest that exposing the following four types of cancer cells (malignant melanoma [B16F1], glioma [F-98], breast adenocarcinoma [MDA-MB-231] and non-small cell lung carcinoma [H1299]) to TTFields for 24 hours resulted in a reduction in cell proliferation compared with untreated cells.6 Combining the findings, Kirson et al concluded that the effect of TTFields were time, frequency and intensity dependent.6 Since then, more studies have been initiated to study the effect of TTFields on different cell lines and animal models. A summary of in vitro studies has been provided below5:
| Type of cancer | Cell line | Frequency, kHz | Intensity, V/cm | Reference |
|---|---|---|---|---|
| High-grade glioma | F-98, C-6, RG-2 | 200 | 1–2 | Cancer Res, 20043 |
| U-118, U-87 | 200 | 1–2 | Proc Natl Acad Sci USA, 20076 Sci Rep, 20157 |
|
| U-87, U-373 | 150 | 0.9 | Oncotarget, 20168 Oncotarget, 20169 |
|
| Ovarian carcinoma | A2780 | 200 | 1.75 | Sci Rep, 20157 |
| A2780, HTB-161, HTB-75 | 200 | 1–2 | Int J Cancer, 201610 | |
| Lung, adenocarcinoma | LLC | 200 | 1–2 | Cancer Res, 20043 |
| A549 | 200 | 1.75 | Sci Rep, 20157 | |
| H1299, A549, HCC827 | 150 | 1.86 ± 0.67 | Semin Oncol, 201411 | |
| Lung, squamous cell carcinoma | HTB-182 | 150 | 1.86 ± 0.67 | Semin Oncol, 201411 |
| Mesothelioma | NCI-H2052, MSTO-211H | 200 | 1.75 | Sci Rep, 20157 |
| Malignant melanoma | B16F10 | 100 | N/A | J Int Med Res, 201212 |
| B16F1 Patricia | 100 | N/A | Cancer Res, 20043 | |
| Breast adenocarcinoma | MCF-7, MDA-MB-231 | 150 | 1.75 | Sci Rep, 20157 Cancer Res, 20043 PloS One, 201513 |
| Cervical cancer | Hela | 200 | 1.75 | Sci Rep, 20157 PloS One, 201513 |
| Pancreatic adenocarcinoma | AsPC-1 | 150 | 1.75 | Sci Rep, 20157 |
| Prostate, adenocarcinoma | PC-3 | 100 | N/A | Cancer Res, 20043 |
| Colorectal, adenocarcinoma | CT-26 | 100 | N/A | Cancer Res, 20043 |
Multiple animal models including rat, mouse and rabbit were used to explore the effect of TTFields on a number of cancer types in vivo. Compared with controls, TTField reduced tumour size in these animal models.5
Summary of in vivo studies of TTFields5:
| Type of cancer | Animal model | Frequency, kHz | Intensity, V/cm | Reference |
|---|---|---|---|---|
| GBM | Rat | 200 | 1–2 | Proc Natl Acad Sci USA, 20076 |
| Ovarian carcinoma | Mouse | 200 | 1–2 | Int J Cancer, 201610 |
| Lung, adenocarcinoma | Mouse | 150 | 1.86 ± 0.67 | Semin Oncol, 201411 |
| Malignant melanoma | Mouse | 100 | N/A | J Int Med Res, 201212 Cancer Res, 20043 Clin Exp Metastasis, 200914 |
| Kidney cancer | Rabbit | 200 | 2.6 ± 0.3 | Clin Exp Metastasis, 200914 |
Clinical data supporting TTFields
In vitro and in vivo findings eventually led to a pilot trial of TTFields, and eventually the prospective, randomized, international phase III EF-11 and EF-14 trials in patients with glioblastoma multiforme (GBM), exploring the therapeutic value of TTFields in cancer treatment.2
For more information about the clinical findings of TTFields, please visit the “Clinical Implications and Safety” section.
Application
TTFields are deployed through two orthogonal pairs of transducer arrays placed on the shaved scalp of patients with GBM or on the upper torso of patients with Malignant Pleural Mesothelioma (MPM), for more than 18 hours a day.2 Maintaining skin integrity plays an important role in treatment compliance and optimizing patient outcome.2
For more information about the application of TTFields device, you may visit the website by scanning the QR code below.

References:
- Mehta M, et al. Crit Rev Oncol Hematol 2017;111:60–65.
- Burri SH, et al. Am J Clin Oncol 2018;41:191–196.
- Kirson ED, et al. Cancer Res 2004;64:3288–3295.
- Swanson KD, et al. Curr Neurol Neurosci Rep 2016;16:8.
- Wang Y, et al. Oncologist 2019;24:e1426–e1436.
- Kirson ED, et al. Proc Natl Acad Sci USA 2007;104:10152–10157.
- Giladi M, et al. Sci Rep 2015;5:18046.
- Kim EH, et al. Oncotarget 2016;7:65125–65136.
- Kim EH, et al. Oncotarget 2016;7:62267–62279.
- Voloshin T, et al. Int J Cancer 2016;139:2850–2858.
- Giladi M, et al. Semin Oncol 2014;41:S35–S41.
- Chen H, et al. J Int Med Res 2012;40:85–94.
- Gera N, et al. PLoS One 2015;10:e0125269.
- Kirson ED, et al. Clin Exp Metastasis 2009;26:633–640.