Platinum agents are the current standard-of-care systemic therapy1:
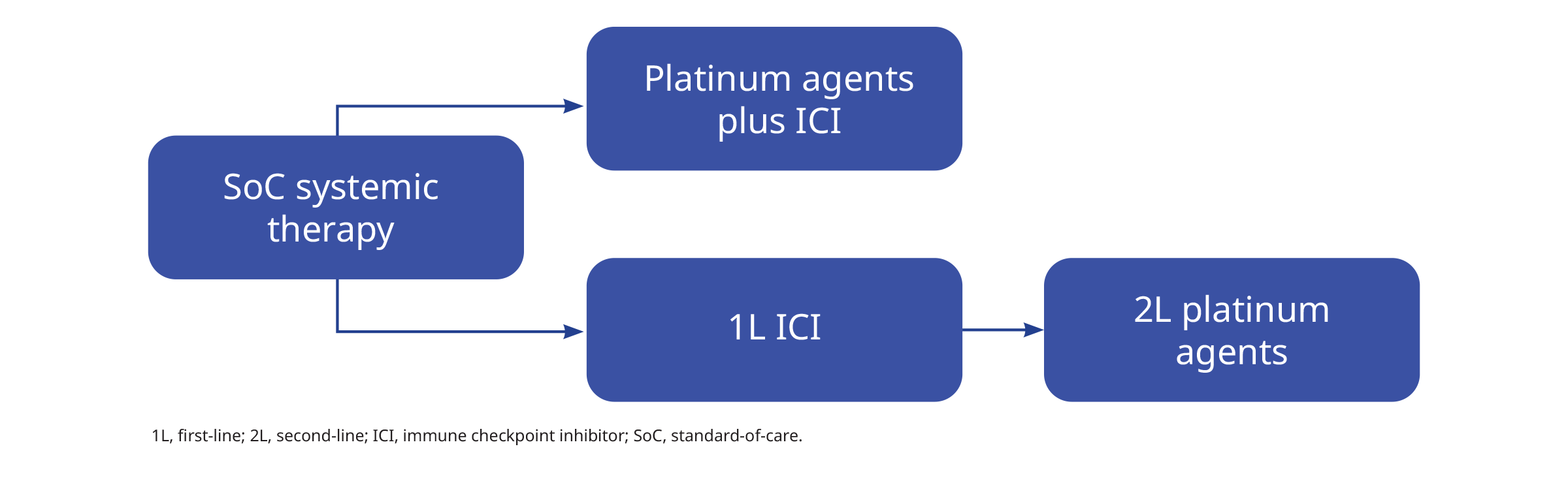
ICI, immune checkpoint inhibitor; SoC, standard-of-care.
A combination of platinum-based chemotherapy plus ICI is the most common first-line treatment for patients with performance status (PS) 0–1, regardless of programmed death-ligand 1 (PD-L1) status.2 These combinations include pembrolizumab plus atezolizumab with or without bevacizumab, nivolumab–ipilimumab, cemiplimab and durvalumab–tremelimumab.2
Single-agent ICI is the first-line treatment for patients with PS 0–1 and high PD-L1 expression (tumour proportion score [TPS] ≥50%).2 In the second- and later-line, chemotherapy could be considered in patients with a PS 0–2 without major comorbidities; a rechallenge with ICI might also be considered if the patient previously benefited from ICI.2
The therapeutic advantage of Tumour Treating Fields (TTFields) in NSCLC was demonstrated in preclinical studies, showing optimal antitumour effects at a frequency of 150 kHz.1 The disruption of mitosis with downstream induction of immunogenic cell death results in an enhanced antitumour immune response.1 Furthermore, TTFields has been shown to amplify the effectiveness of ICI or taxanes in preclinical models, thus supporting the combination of TTFields and ICI.1
A phase 1/2 trial demonstrated the safety and feasibility of TTFields in combination with pemetrexed as a second-line therapy for patients with advanced NSCLC.3 Based on these findings, the pivotal phase 3 LUNAR study was conducted to evaluate the safety and efficacy of TTFields in combination with standard systemic therapies for patients with metastatic NSCLC progressing on or after platinum-based therapy.1
References:
- Leal T, et al. Lancet Oncol 2023;24:1002-1017.
- Hendriks LE, et al. Ann Oncol 2023;34:358-376.
- Pless M, et al. Lung Cancer 2013;81:445-450.
LUNAR in metastatic NSCLC
The LUNAR study was a randomised, open-label, pivotal phase 3 study investigating the efficacy and safety of TTFields in combination with standard systemic therapy (ICI or docetaxel) in adult patients with NSCLC following progression on or after platinum-based therapy.1
Study design2
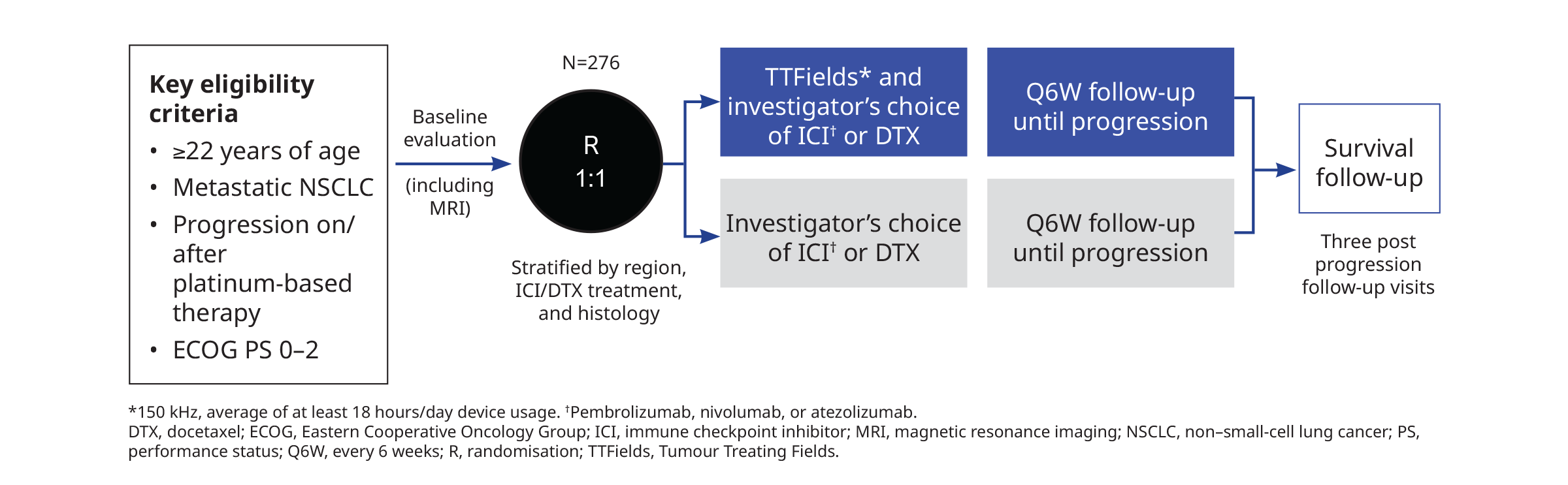
- Primary endpoint: Overall survival (OS)
- Key secondary endpoints: OS in the docetaxel and ICI subgroups
- Other secondary endpoints:
- Progression-free survival (PFS)
- Overall response rate (ORR)
- OS by squamous and non-squamous histology
- Patient-reported, health-related quality-of-life (HRQoL) scores
- The study recruited 276 patients at 130 sites in 19 countries aged 22 years and above with metastatic NSCLC progressing on or after platinum-based therapy, with squamous or non-squamous histology and ECOG performance status of 2 or less.1 No restriction was placed on the number or type of prior lines of systemic therapy.1 Patients were randomised at a 1:1 ratio to receive TTFields with standard therapy (n=137) or standard therapy alone (n=139).1 Systemic therapies were given according to local practice guidelines whereas TTFields of 150 kHz was delivered continuously to the thoracic region to achieve an average of at least 18 hours per day device usage.1
Baseline characteristics
About one-third (32%) of patients had received a previous IC and 87% of patients received one previous line of systemic therapy.1 The baseline characteristics of the study population are summarised below:
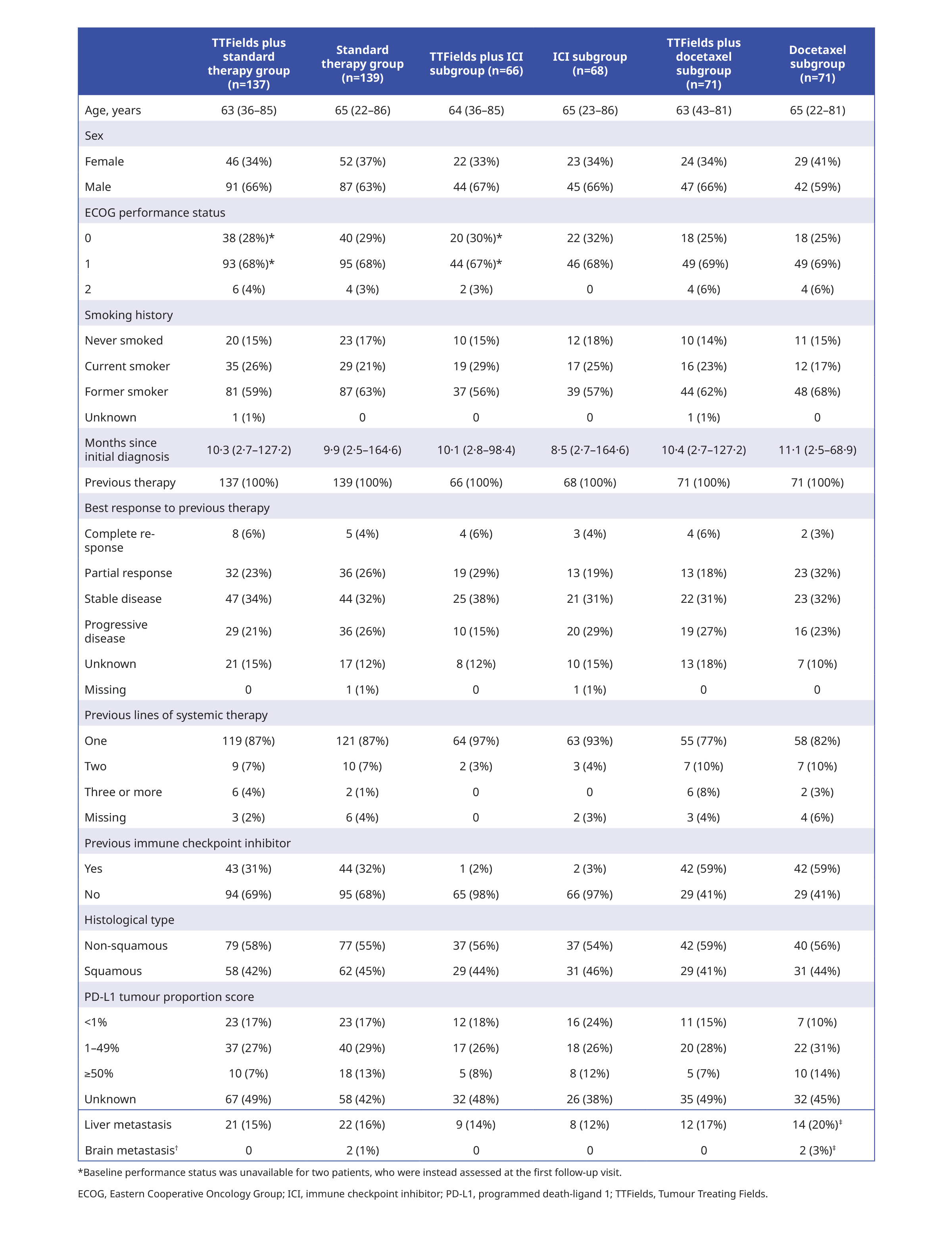
Efficacy
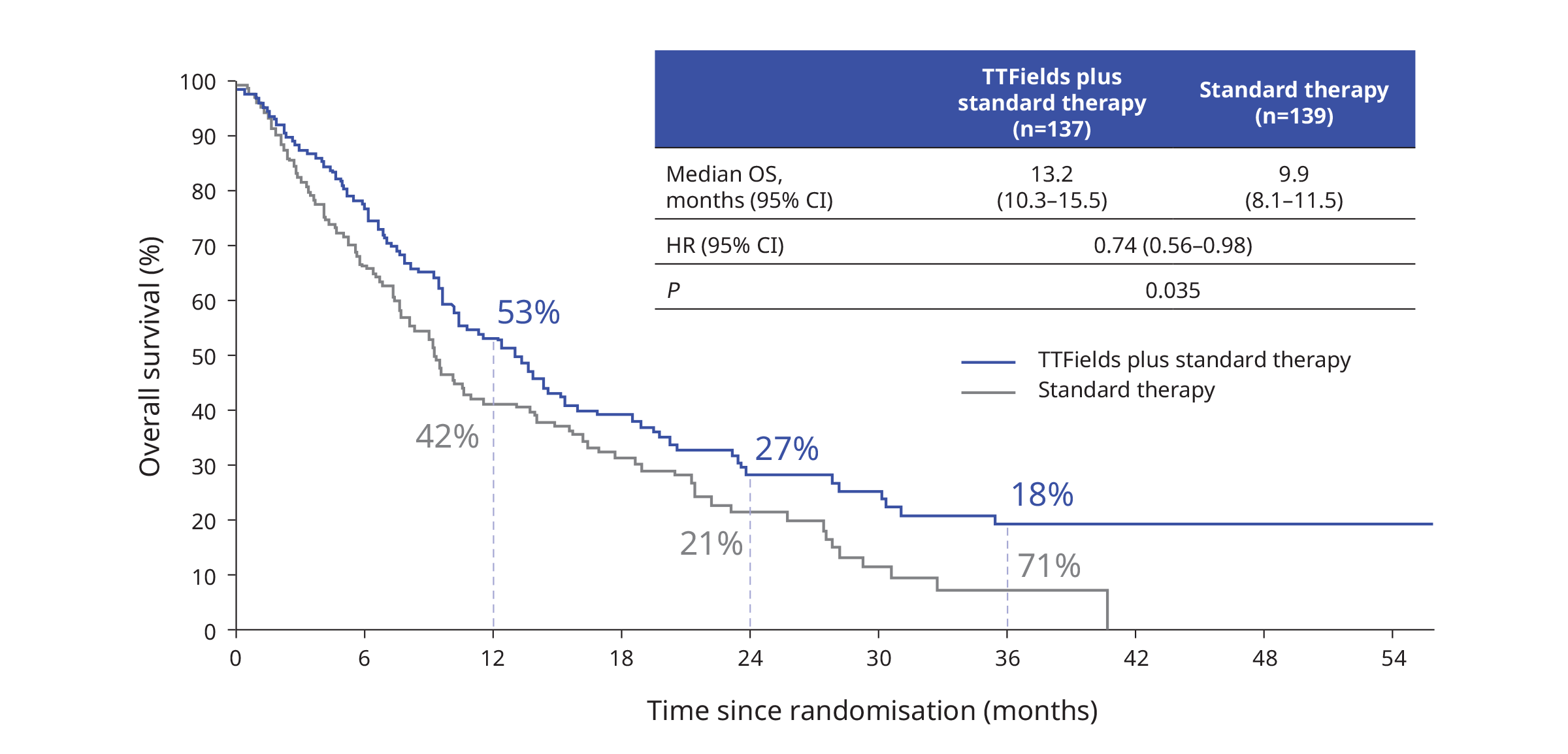
The primary endpoint was successfully reached with significantly longer OS in patients receiving TTFields plus standard therapy versus those receiving standard therapy alone1:
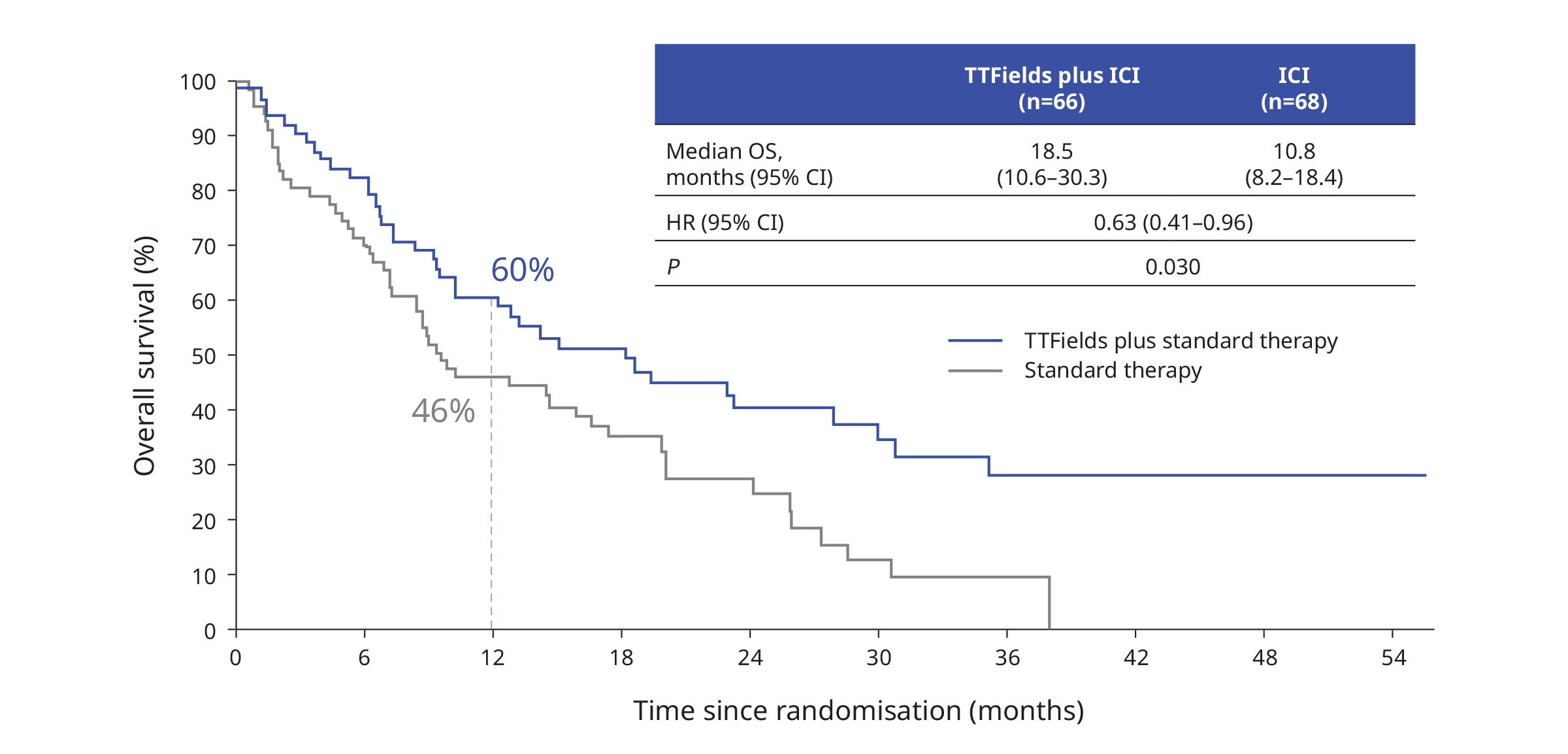
OS was improved significantly with the addition of TTFields in the ICI subgroup1:
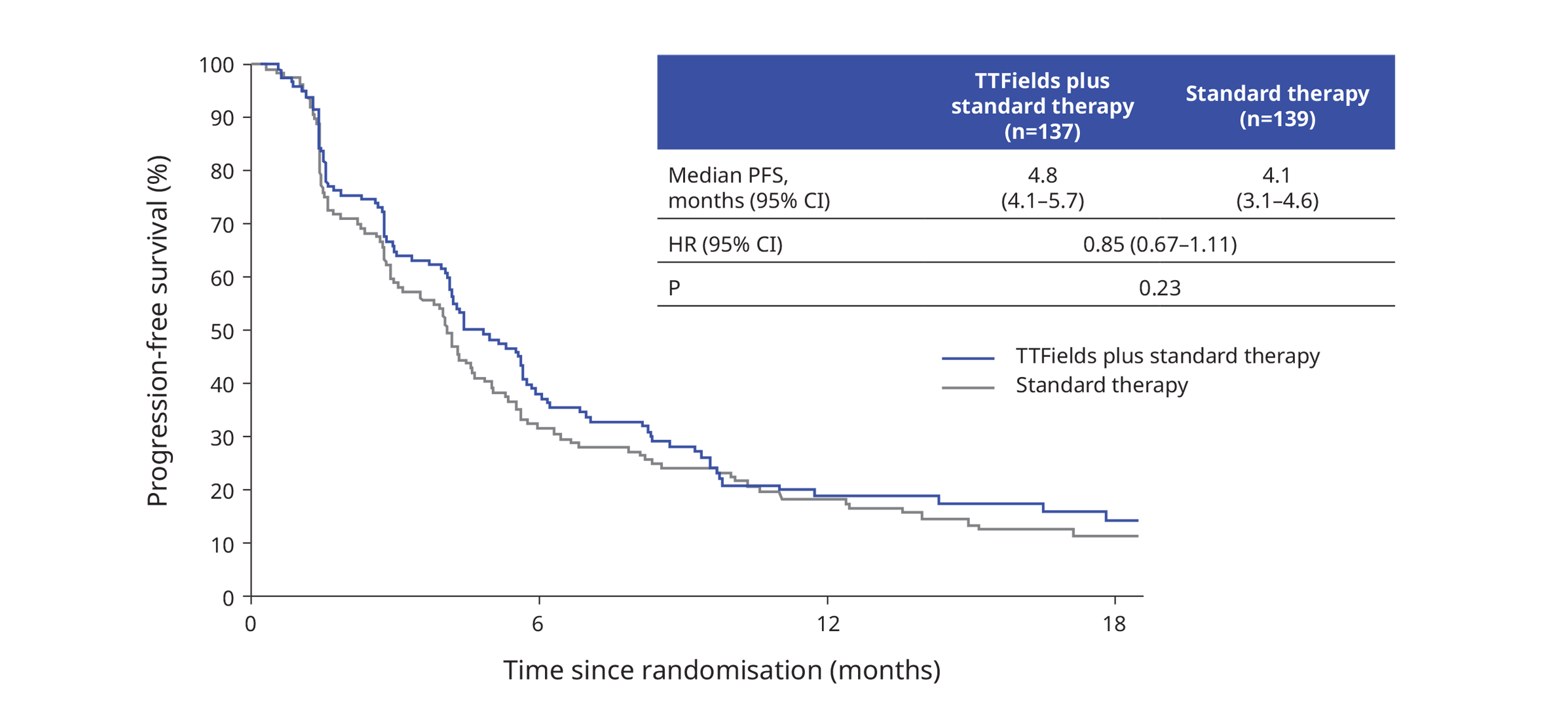
PFS was similar for patients receiving TTFields plus standard therapy versus standard therapy alone1:
The ORR of TTFields and standard therapy was 20.4% (95% CI 14.0–28.2) versus 17.3% (11.4–24.6) with standard therapy alone (two-sided p=0.50).1 All complete responses (n=5) occurred in patients receiving an ICI, of which four received TTFields in combination with ICI and one received only ICI.1
Histology did not significantly affect OS1:

Patient-reported global health status, measured by EORTC QLQ-C30 questionnaire, did not decline in both treatment arms over 54 weeks of follow-up, and there was no significant difference between treatment groups.1
Safety
Dose reductions to the standard therapy regimen were required in 12% of patients receiving TTFields with standard therapy and 14% of the group receiving standard therapy alone.1 Overall, 11% of patients discontinued standard therapy due to toxicity related to the standard therapy.1 In patients who received TTFields, 14% discontinued due to toxicity related to device usage.1
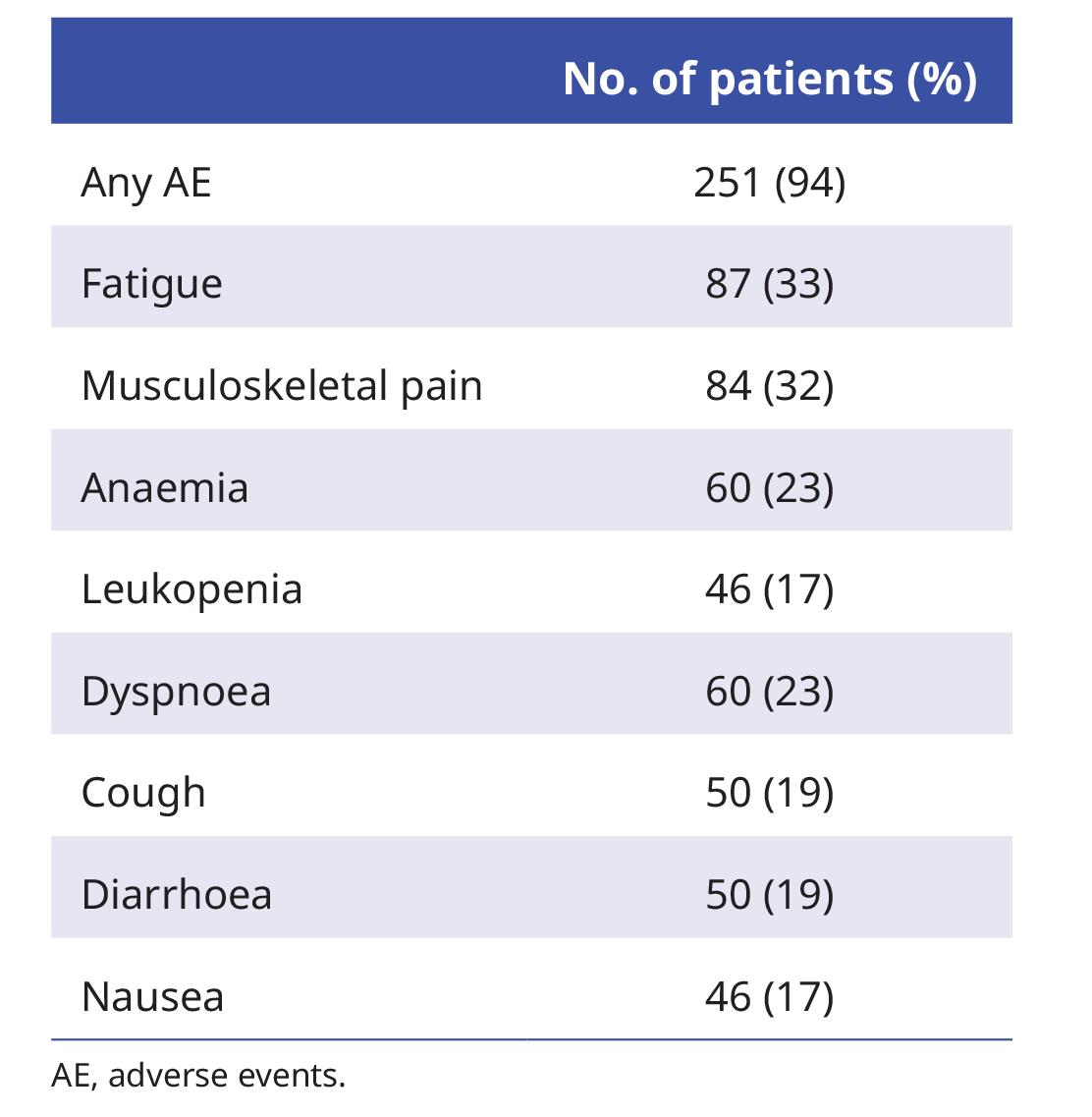
Apart from dermatitis (22%), the most frequently reported adverse events (AE) were associated with the systemic therapies or the underlying cancer1:
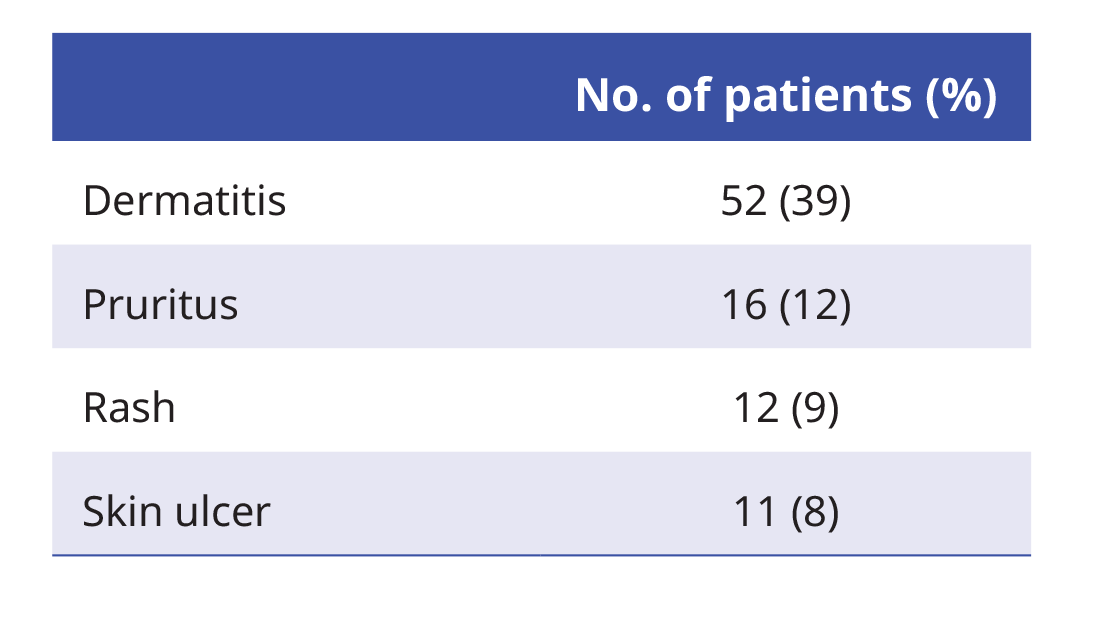
At least one device-related AE was reported in 71% of patients receiving TTFields.1 The most frequent TTFields-related events were grade 1–2 skin AE1:
Post hoc exploratory analysis on efficacy by PD-L1 status in the ICI subgroup
The survival benefit of TTFields plus ICI appears to be more pronounced in patients with PD-L1 (TPS ≥1%) tumours2:
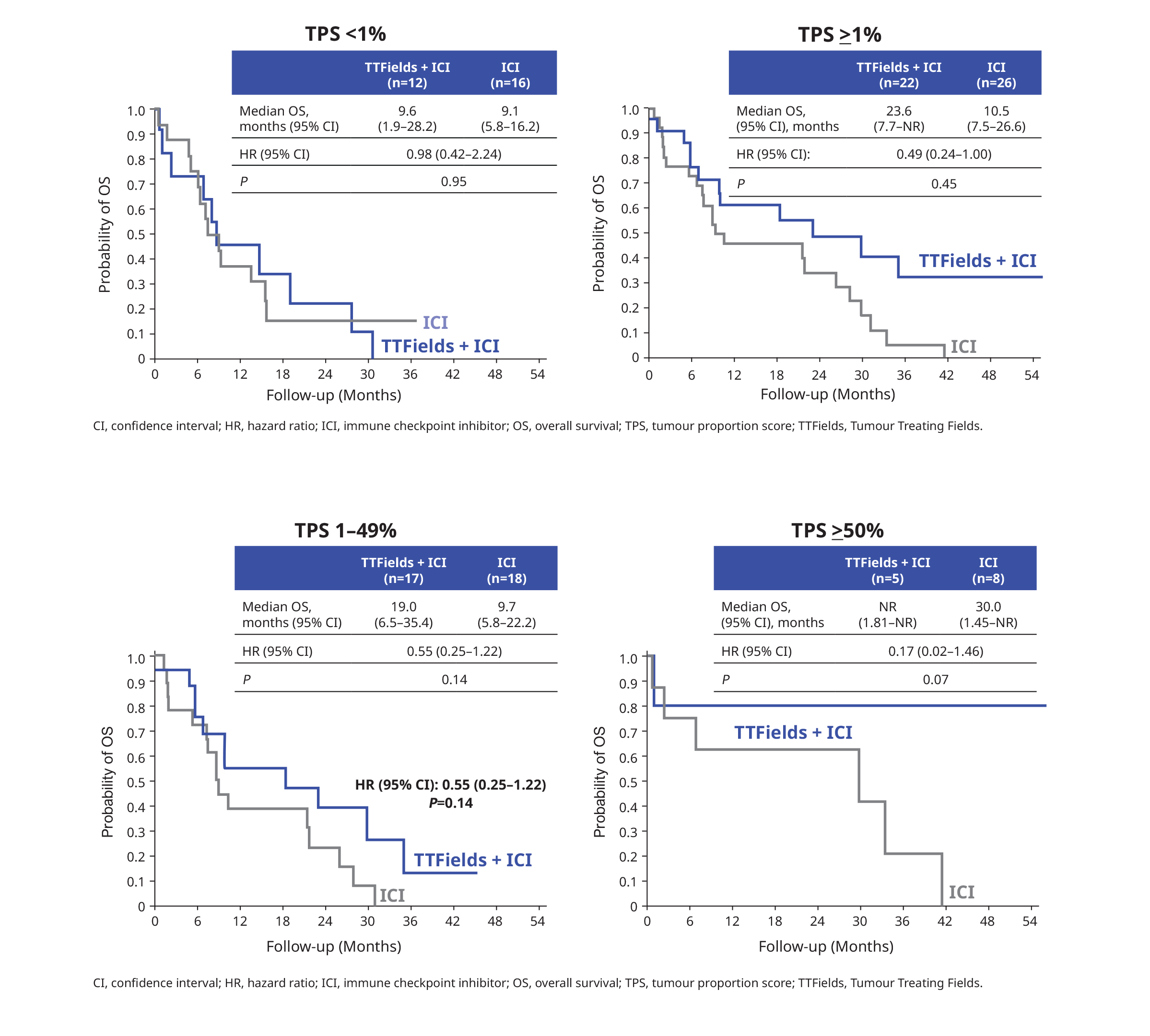
High PD-L1 expression was associated with improved survival.2 However, more evidence will be required due to the small sample size and the exploratory nature of this analysis2:

Summary
Overall, the phase 3 LUNAR study has shown promising potentiating effect of TTFields for ICI.3 These pivotal efficacy and safety data suggest TTFields as a potential treatment option in managing metastatic NSCLC in later-line setting.1
References:
- Leal T, et al. Lancet Oncol 2023;24:1002-1017.
- Leal T, et al. TTFields and immune checkpoint inhibitor in metastatic non–small-cell lung cancer: PD-L1 subgroups in the phase 3 LUNAR study. Presented at: 2023 World Conference on Lung Cancer; 9–12 September 2023; Singapore. Abstract OA22.05.
- Fennell DA. Lancet Oncol 2023;24:946-947.